Welcome to the monthly SHDA Research Update. Our specialists have selected 3 papers that have been published recently in each speciality (cardiology, cardiothoracic surgery, cardiac imaging, perioperative medicine).
Cardiology
Gupta et al. ENGAGE AF–IMI 48 Echocardiographic Study Investigators. The Prognostic Significance of Cardiac Structure and Function in Atrial Fibrillation: The ENGAGE AF-TIMI 48 Echocardiographic Substudy. J Am Soc Echocardiogr. 2016 Jun;29(6):537-44.
Although atrial fibrillation (AF) is associated with thromboembolism there is a lack of knowledge about the relationship between cardiac structure and risk of thromboembolism. This sub-study of the ENGAGE AF-IMI 48 trial involved transthoracic echocardiography of 971 subjects with non-valvular AF and CHADS2 ≥ 2. After a median of 2.5 years follow-up there were 48 (4.9%) recorded thromboembolisms and 89 (9.2%) deaths. Statistical modelling to adjust for confounding factors, revealed two echocardiographic parameters were significantly and independently associated with increased risk of death: larger left ventricular (LV) end-diastolic volume index (LVEDVI per 12.9 ml/m2; HR 1.49; 95% CI 1.16–1.91) and higher LV filling pressures (E/e’ ratio per 4.6 ml/m2; HR 1.32; 95% CI 1.08–1.61).
The authors concluded that:
“LV size and filling pressures may help identify patients with AF at increased risk for death and could be targets for therapies directed at improving survival.”
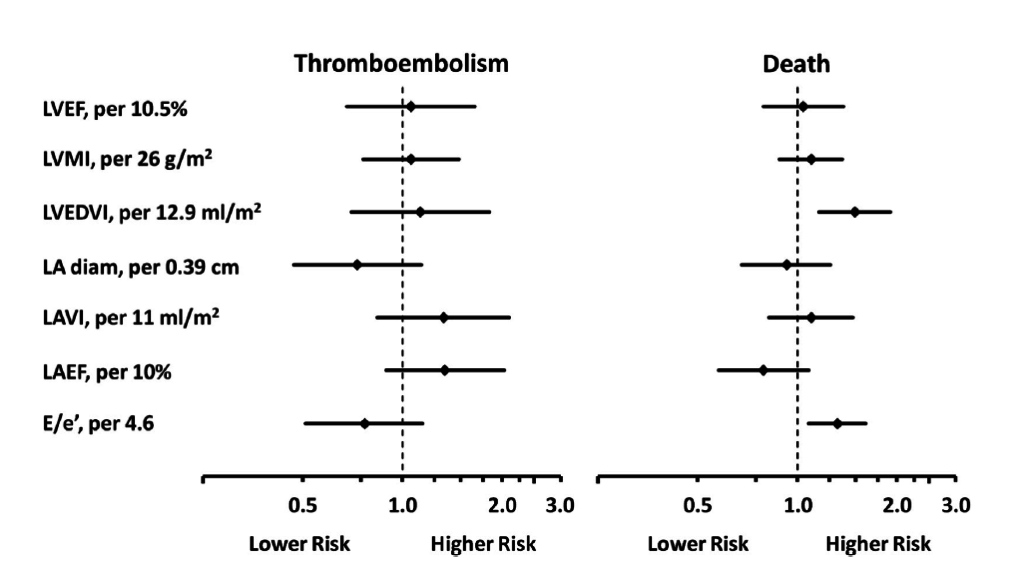
Figure: Forest plot showing echocardiographic parameters and risk of thromboembolism or death.
In this longitudinal study the development of hypertrophic cardiomyopathy was investigated in 38 sarcomere mutation carriers (mean age 15.8 years) without baseline left ventricular hypertrophy (LVH). Participants were followed for a median of 2.9 years and underwent imaging and biomarker analysis; four (10%) developed hypertrophic cardiomyopathy. The development of LVH (i.e., development of maximal LV wall thickness ≥ 12 mm in adults or z-score ≥ 3 in children < 18 years old) was monitored using echocardiography. Significant differences were observed in sarcomere mutation carriers who developed HCM for parameters including: higher LV ejection fraction (74% vs 69%; P = 0.02); lower global E’ velocity (11.2 vs 14.8 cm/s; P < 0.0001); longer mitral valve posterior leaflet length; and higher baseline serum NT-proBNP values (208 vs 57 pg/mL; P = 0.04). It was also observed that two (50%) of the participants who developed HCM had ≥1 mm ST segment depression at baseline ECG.
“LV relaxation, ECG changes, mitral leaflet length, and serum NT-proBNP concentrations appeared more prominently abnormal at baseline in preclinical sarcomere mutation carriers who imminently progressed to HCM,” the researchers concluded.
This manuscript presents the results of the ENSURE-AF study, a phase 3b prospective randomised trial. The primary aim of this trial was to determine the efficacy and safety of edoxaban (a non-vitamin K antagonist oral anticoagulant or NOAC) compared with enoxaparin-warfarin in non-valvular AF patients undergoing cardioversion.
Efficacy was compared using a composite endpoint of stroke, systemic embolic event, myocardial infarction, and cardiovascular mortality and safety using a composite endpoint of major and clinically relevant non-major (CRNM) bleeding. In total 1,095 patients were randomised to receive edoxaban and 1,104 to receive enoxaparin-warfarin, with 90% and 88% cardioverted respectively. The efficacy endpoint occurred in 5 patients in the edoxaban group versus 11 in the enoxaparin–warfarin group (<1% vs 1%; OR 0.46; 95% CI 0.12, 1.43) and the safety endpoint occurred in 16 patients in the edoxaban group vs 11 in the enoxaparin group (OR 1.48; 95% CI 0.64, 3.55).
The authors concluded that:
“Edoxaban is an effective and safe alternative to the best possible conventional treatment with enoxaparin and vitamin K antagonist strategy, and might allow prompt cardioversion to be performed.
Cardiac Surgery – Summarised by Andrew Haymet
Aortic valve sparing (AVS) procedures, such as reimplantation of the aortic valve and remodelling of the aortic root, have expanded the range of surgical treatment options for patients with aortic root dilatation. Aortic valve reimplantation is most appropriate for young adults with aortic root aneurysms, whilst remodelling of the aortic root is best suited to older patients with ascending aortic aneurysms or AI secondary to dilatation of the sinotubular junction, with a normal aortic annulus and cusps. Remodelling encompasses sinotubular junction adjustment up to replacement of all three aortic sinuses. AVS procedures are sophisticated and require a minimum case volume to build and maintain technical expertise.
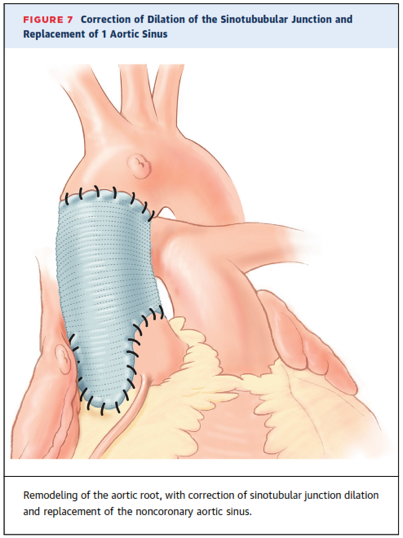
Figure: Remodelling of the aortic root, with correction of sinotubular junction dilatation and replacement of the noncoronary aortic sinus.
The optimal surgical management of coarctation of the aorta with associated intracardiac defects remains controversial. 31 patients received a novel, single stage repair technique for this condition using a midline sternotomy. Of these, 28 had associated distal arch hypoplasia and one had a type 1 interruption. The coarctation and, if applicable, the distal arch, were repaired prior to the institution of cardiopulmonary bypass (CPB), which was then used for repair of intracardiac defects. All patients had successful repair of coarctation and distal arch hypoplasia without the need for rescue CPB. Fulminant sepsis accounted for one in-hospital mortality (3.22%). There was no late mortality. Overall event-free survival and intervention-free survival at 2 and 3 years was 88.5% and 91.7% respectively. The authors concluded this technique was feasible “without the use of hypothermic circulatory arrest or regional cerebral perfusion, with acceptable and reproducible results.”
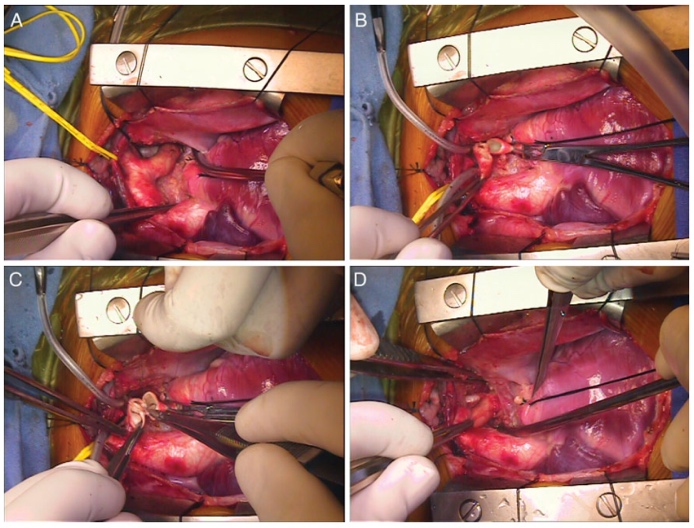
Figure: (A) Intraoperative photograph of proximal arch and coarct segment. The pulmonary artery is retracted inferiorly and to the right. (B) Proximal and distal cut ends of aorta after excision of coarct segment. Note placement of clamp on descending aorta. (C) Tension-free posterior anastomosis. (D) Completed anastomosis.
An off pump, endovascular approach to treat severe mitral valve regurgitation would benefit a substantial number of patients who are ineligible for conventional surgery. Twenty-two pigs received a self-expanding mitral valved stent in an off-pump procedure. One group (AP; n=12) received an apical fixation technique and served as control, with the other receiving subannular fixation (SA; n=10). Paravalvular leakages were trivial or less in 19 of 20 cases. The mean gradients across the valved stent increased but remained within physiological ranges (AP: 1.2 ± 0.6 mmHg, SA: 2.6 ± 2.0 mmHg). The ejection fraction decreased after stent deployment (AP: 52 ± 10%, SA: 48 ± 4%), with a greater mean survival time in the AP group over SA. Proof of principle was established with stable haemodynamics, acceptable gradients and physiological longitudinal function. Longer term outcomes were superior in the apical fixation group over the subannular group.
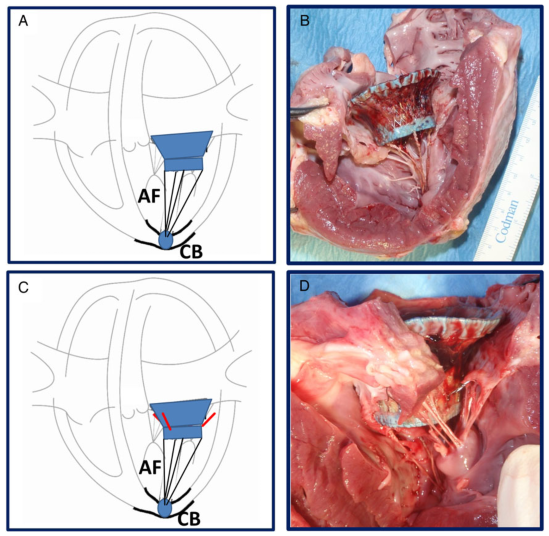
Figure: Schematic drawing and gross-evaluation image of (A and B) the apical fixation (AF) system consisting of four individual neochords affixed against an apical, button-like counter bearing (CB) and (C and D) the design with subannular fixation elements to be positioned directly below the mitral annulus and additional apical fixation.
Cardiac Imaging
This study of 102 consecutive patients with newly diagnosed left ventricular (LV) dysfunction and atrial tachyarrhythmias investigated the prediction of tachycardia-induced cardiomyopathy using cardiac magnetic resonance imaging.
TIC was diagnosed for patients whose LV ejection fraction improved to ≥50% during a 1-year follow-up and dilated cardiomyopathy (DCM) was diagnosed if the LV ejection fraction did not improve. CMR showed that TIC patients had significantly lower RVEF/LVEF ratios and higher RVEDV/LVEDV ratios compared with DCM patients. In the multivariate analysis and RVEF/LVEF ratio less than 1.05 was the best predictor of TIC (sensitivity 69.1%; specificity of 91.5%).
“Among patients with newly diagnosed LV dysfunction and atrial tachyarrhythmias, CMR analysis revealed depressed RV systolic function and dilatation in TIC patients, and the presence of RV dysfunction had a high predictive value for TIC,” the researchers concluded.
In this study patients with arrhythmic mitral valve prolapse (MVP) (36 with LV late gadolinium enhancement on CMR and no or trivial mitral regurgitation and 16 without LV late gadolinium enhancement) were investigated by morphofunctional CMR. The researchers hypothesized that a localized mechanical stretch on the LV myocardium by the prolapsing leaflets produces replacement-type fibrosis. It was observed that, in MVP patients with late gadolinium enhancement, the following parameters were higher: mitral annulus disjunction, end-systolic mitral annular diameters and end-diastolic mitral annular diameters, prevalence of posterior systolic curling, and basal to mid LV wall thickness ratio. Furthermore, a linear correlation was found between mitral annulus disjunction and curling (R = 0.85) and there was a higher prevalence of auscultatory midsystolic click in the patients with late gadolinium enhancement.
The authors concluded that:
“The present study clearly shows that MAD and systolic curling of the posterior MV leaflet are associated with LV fibrosis, accounting for the excessive mobility of the MV apparatus and systolic stretch of the myocardium closely linked to the valve.”
In a retrospective study echocardiographic predictors of outcome were investigated in 169 consecutive patients with severe functional mitral regurgitation (FMR) who were undergoing percutaneous mitral valve repair with MitraClip. The primary end-point was cardiovascular mortality and at three years, survival free of cardiac death was 61.6%. Statistical analysis revealed that independent predictors of cardiovascular mortality were severely impaired renal function (GFR < 30 ml/min; OR = 5.46, 95% CI 1.43, 20.84; P = 0.01) and RV systolic dysfunction PSVtdi < 9.5 cm/s (OR = 0.57; 95% CI 0.39, 0.82; P = 0.003).
“Our study shows the importance of RV systolic function evaluation for the risk stratification of patients with FMR and heart failure undergoing MitraClip therapy,” the researchers concluded.
Perioperative Medicine – Summarised by Andrew Haymet
Mechanical prosthetic valve thrombosis (PVT) is a potentially catastrophic event, with a high mortality attributable to late presentations of advanced heart failure symptoms. Surgical risk is high, with adverse perioperative outcomes proportional to worsening NYHA functional class on presentation and decreasing haemodynamic stability. This unique case describes an unstable patient with multiorgan failure who required an urgent fourth redo sternotomy, cardiotomy and valve replacement. The use of VA-ECMO enabled resolution of hepatic dysfunction and coagulopathy, and avoided the need for renal replacement therapy. Extracorporeal life support may provide a means for optimizing haemodynamics in multiorgan dysfunction and high perioperative risk prior to definitive surgical intervention.
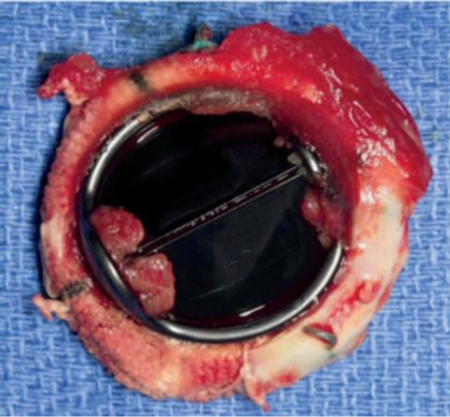
Figure: Valve thrombosis, ventricular side.
Mitral regurgitation is one of the most prevalent valvular lesions globally. Moderate to severe MR increases preload and reduces afterload, chronically altering LV loading and potentially inducing adverse ventricular remodeling. Percutaneous mitral valve repair using MitraClip is emerging as a sound alternative for patients of unacceptably high risk for conventional mitral repair or replacement. Unfortunately, Mitraclip therapy may acutely trigger afterload mismatch, especially in patients with severe baseline LV systolic dysfunction. Prompt diagnosis and treatment with echocardiography and inodilator support, instead of vasopressors, may influence patient outcomes. Anaesthetists should be aware of this perioperative complication and plan pre-emptively where possible.
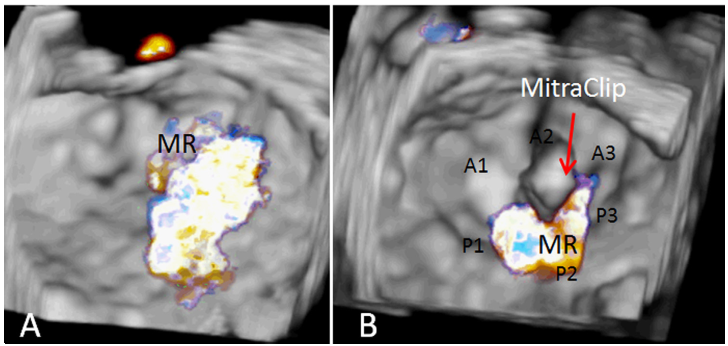
Figure: (A) 3-dimensional transesophageal echocardiography (TEE) en-face view of the mitral valve with color-flow Doppler demonstrating moderate-to-severe mitral regurgitation (MR) due to degenerative mitral valve disease. (B) Three-dimensional TEE en-face view of the mitral valve with color-flow Doppler after successful MitraClip repair demonstrating mild MR. A1, A2, and A3 represent segments of the anterior mitral valve leaflet; P1, P2, and P3 represent the scallops of the posterior mitral valve leaflet.
Hypertrophic cardiomyopathy (HCM) has a prevalence of 0.2% and is one of the leading causes of sudden cardiac death in the young. Atrial fibrillation (AF) is the most common arrhythmia in patients with HCM, affecting 20%, and this combination confers substantially higher risk of stroke, heart failure and mortality. Patients with AF should be anticoagulated, with a subset of patients with limiting symptoms being eligible for rhythm control strategies such as amiodarone, disopyramide or catheter ablation. Further research is required to refine detection of AF in patients with HCM and mitigate its associated sequelae.
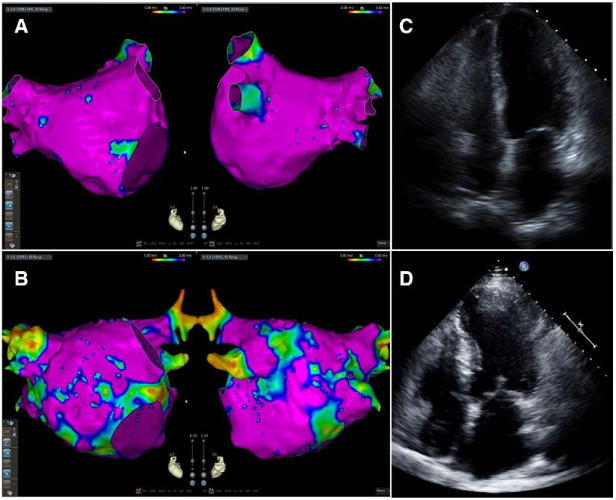
Figure. Pathological left atrial electromechanical remodelling underlies relatively low success rates with catheter ablation of atrial fibrillation (AF) in hypertrophic cardiomyopathy (HCM). Electroanatomic bipolar voltage map in the anterior-posterior and posterior-anterior views of (A) a patient with paroxysmal AF and a structurally normal heart and (B) a patient with persistent AF and HCM. Low voltage (<0.4mV) suggesting scar is shown in red. Healthy tissue is shown in pink. Note the increased scar burden in the patient with HCM and persistent AF. Corresponding apical 4-chamber echocardiographic images reveal normal left atrial (LA) volume index (LAVI; 29.4 mL/m) in the patient with paroxysmal AF (C) and severe LA dilation (LAVI=65.7 mL/m) in the patient with HCM and persistent AF (D).